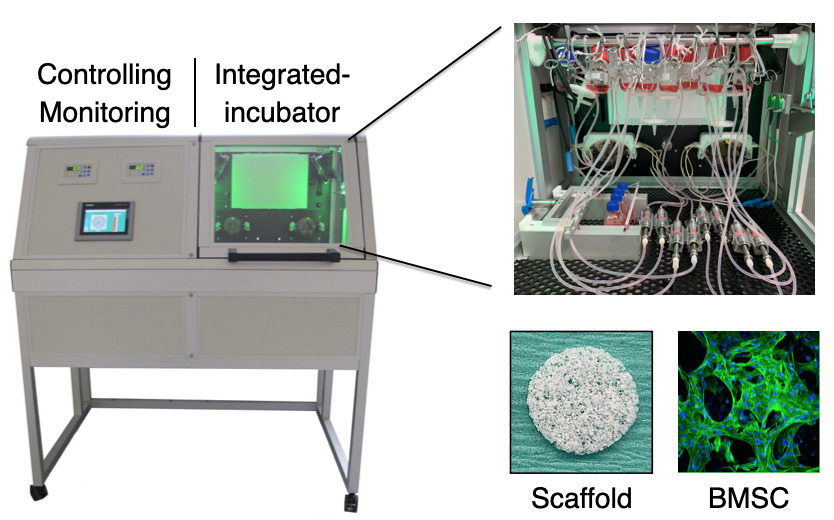
Optimization of capacity and cost of ex-vivo expansion using bioreactors to facilitate commercialization of MSC as ATMP
The fate and growth of mesenchymal stem cells are regulated not only by bioactive molecules but mechanical stimuli. A number of bioreactor systems that create dynamic cell culture environment have been developed by aiming at improving cell growth and targeted differentiation.
In the project, a custom-designed perfusion bioreactor has been utilized to evaluate the effect of fluid stimuli on bone marrow mesenchymal stem cell (BMSC) fate and growth on 3D polymeric scaffolds.
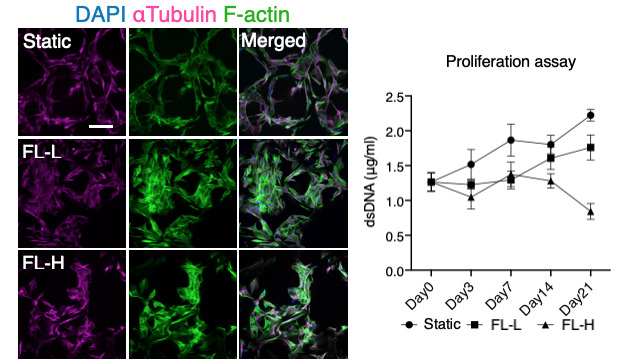
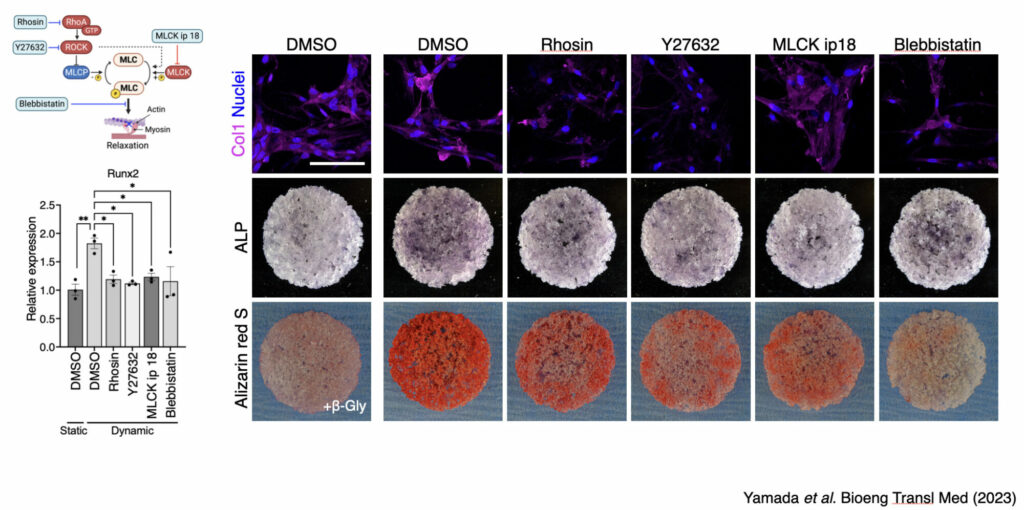
Even under the effect of sub-physiological level of fluid stimuli, we found that the formation of filamentous actin of BMSC was highly enhanced, which was associated with decreased cell proliferation.
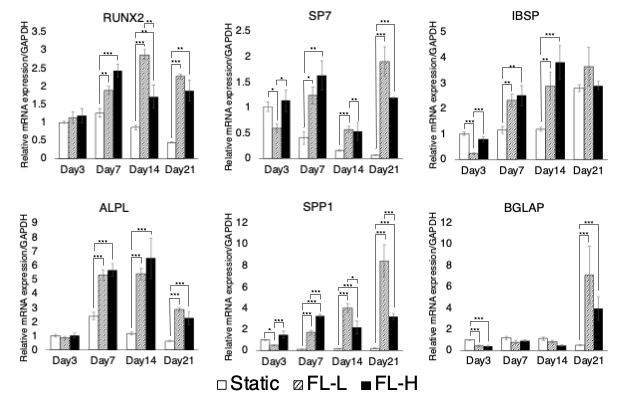
Contrarily, fluid flow stimuli induced the initiation of osteogenic differentiation confirmed by multiple assays including mRNA expression of osteogenic markers and collagen matrix formation.
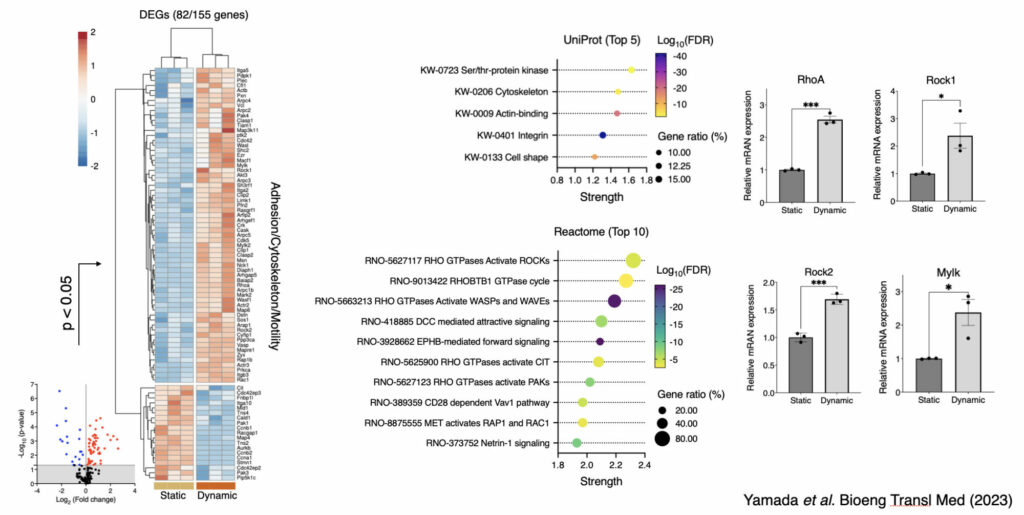
In addition, molecular mechanism behind mechanically induced osteogenic differentiation was thoroughly assessed, and key signaling pathway was identified.
Under the dynamic environment, BMSC activated Rho-ROCK signaling pathway, which is crucial for actomyosin-inducing cell contractility.
However, when the cell contractility is mitigated by cell contraction inhibitors, osteogenic phenotype and functionality disappeared. There findings open the possibility of preconditioning BMSC to be of more osteogenic phenotype by aiming at improving regenerative efficiency after cell transplantation therapy.
Current projects also include the investigation of mechanotransduction related to flow-inducing osteogenesis, further optimization of ideal flow magnitude for osteoinduction and developing next generation bioreactor systems.