Reconstruction of jaw bones using stem cells and biomaterials
Bone loss in craniofacial region is caused by various factors including inflammation, injury, neoplastic diseases and congenital diseases. Due to the limited capacity of spontaneous healing, treatment of bone defects often requires extensive surgical intervention. Current therapuetic golden standard to such defects is autologous bone graft, but complete regeneration is scarcely achieved despite great burdens on the patients.
Alternatively, tissue engineering approach, namely the combination of stem cells, biomaterials and bioactive molecules, has emerged in regenerative medicine. The aim of the clinical trial was to evaluate the safety and therapeutic efficacy of stem cell therapy for bone regeneration in oral region.
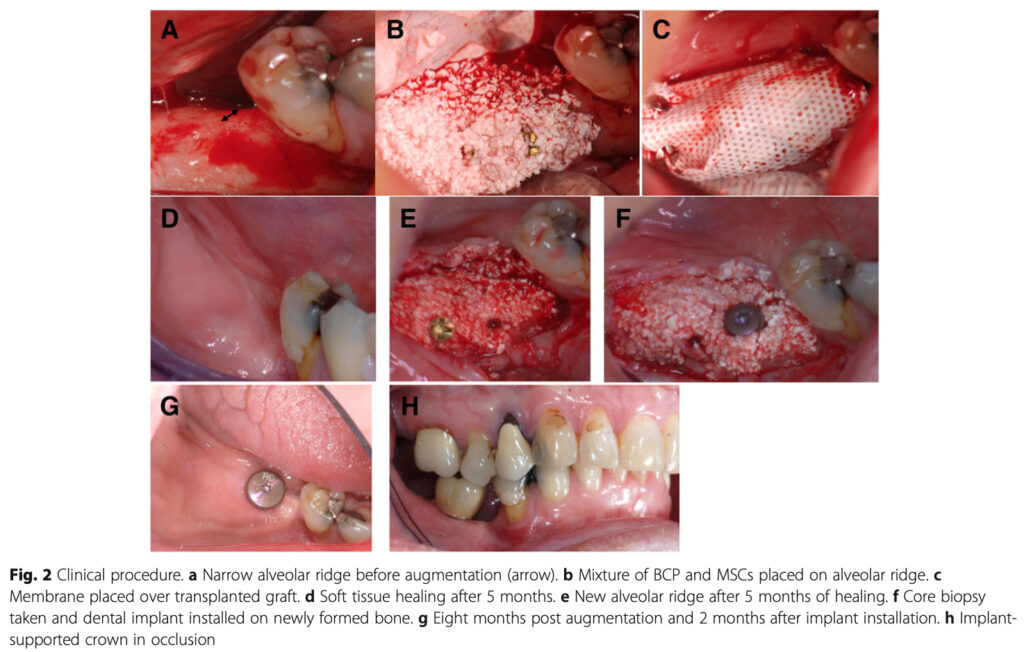
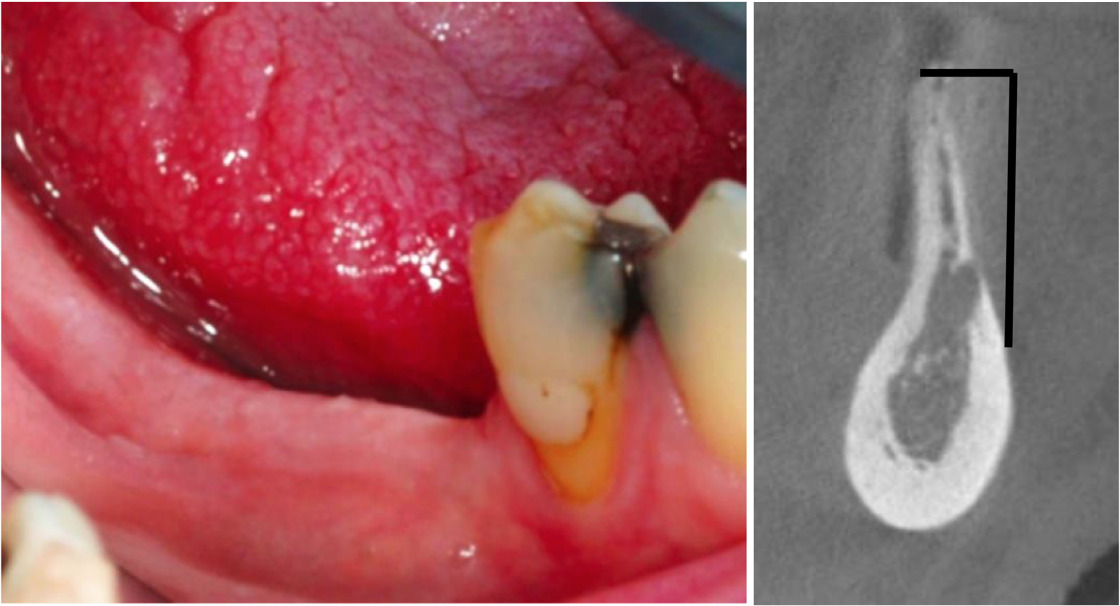
Patients with atrophied alveolar ridge were voluntarily recruited. Bone marrow mesenchymal stem cells (BMSC) were harvested from the patient and then expanded in accordance with cGMP in prior to cell transplantation.
BMSC were seeded on BCP granules and transplanted into an area where dental implant had been planned to be placed. Clinically, after 5 months of healing period, bone regeneration was achieved, and dental implant was successfully placed on the donor site with decent initial stability.
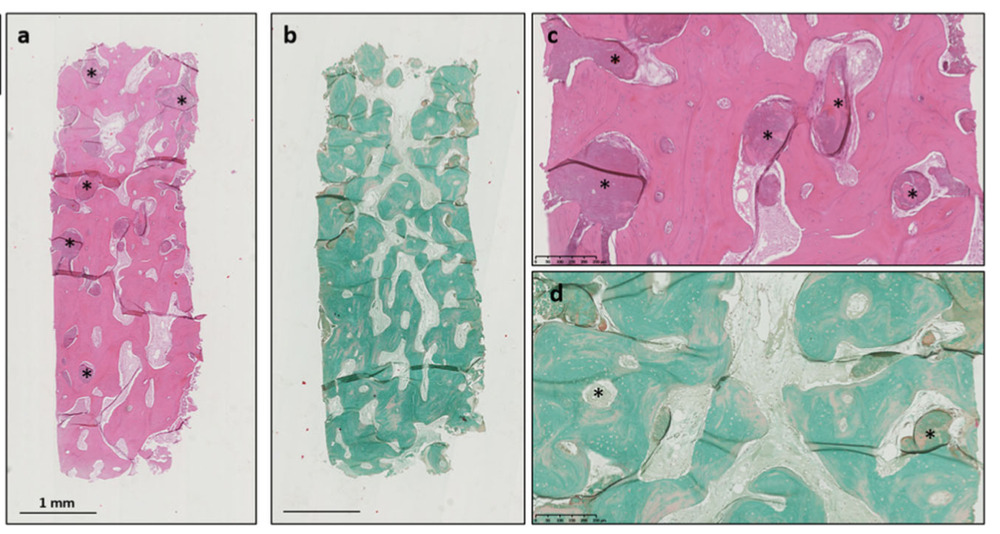
Histologically, a significant amount of bony tissues were generated at the donor site without a sign of inflammatory and atrophic tissue formation.
The clinical trial confirmed the safety of the transplantation therapy and therapeutic potential in severely atrophied bony defects.
Currently, the optimization of biomaterials ideal for scaffoldings is being undertaken including material development and biological evaluation. Furthermore, based on these results, the large-scale randamized clinical trial is being implemented.
Reference
Gjerde, C., Mustafa, K., Hellem, S. et al. Cell therapy induced regeneration of severely atrophied mandibular bone in a clinical trial. Stem Cell Res Ther 9, 213 (2018)